Ion Activity
The activity of an ion is its ‘effective’ concentration in solution. The reason that the effective concentration of an ion is different than the actual concentration (as measured in a water sample) is because ions form uncharged pairs in solution (see Figure 1). The extent of ion-pair formation increases with salinity. There is little difference between the activities and concentrations of most ions at salinty’s below about 3 ppt.
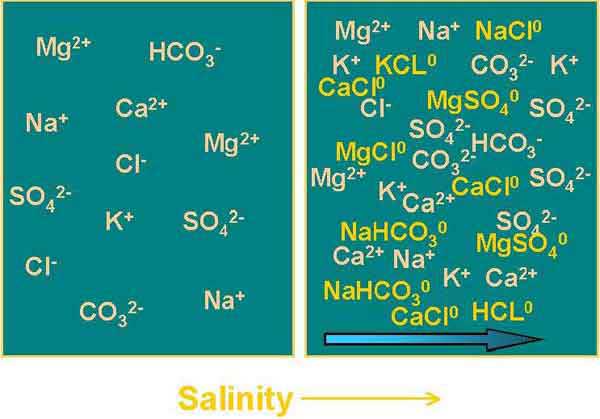
Figure 1. The extent of uncharged (designated with ‘o’) ion pair formation increases with salinity, and this reduces the ‘effective’ concentration of ions.
There are several software packages that can be used to to calculate activity coefficients for ions based on Debeye-Huckel theory or Pitzer equations. The Pitzer approach is most suitable when salinity’s are in the seawater range.
« Back to Glossary Index