Water Hardness
Water �hardness’ is defined as the content of metallic ions in waters which react with sodium in soaps. Most water hardness is attributable to calcium (Ca2+) and magnesium (Mg2+) ions. A general expression used to calculate water hardness is:
hardness = 2.5(Ca2+) + 4.1(Mg2+)
in which calcium and magnesium are measured in milligrams per litre (mg L-1) [1]. Water is considered very hard if hardness values exceed 150 mg L-1 [1]. In comparison, hardness values of < 60 mg L-1 are indicative of soft waters [1]. Seawater has a hardness level of 500 mg L-1. Water hardness increases with salinity in a mixing system between seawater and rainwater/river water (Figure 1).
Some important mineral sources of calcium and magnesium ions are calcium carbonate (CaCO3) and dolomite (CaMg(CO3)2) dissolved from limestones.
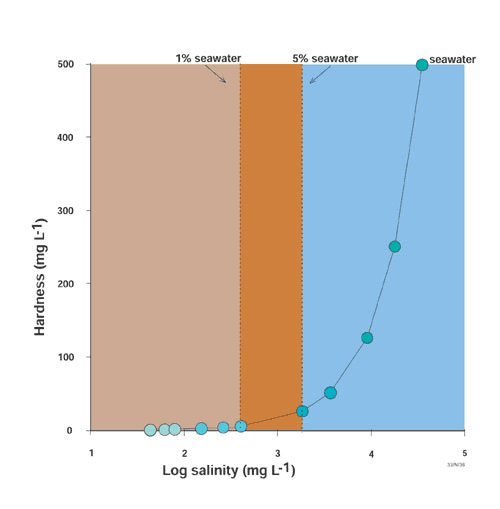
Figure 1. Changes in water hardness with log salinity in a mixing system between seawater and rainwater.
- Freeze, R.A. and Cherry, J.A. 1979. Groundwater, Prentice-Hall Inc., Englewood Cliffs, N.J., pp. 604.